ReO2 Contact Lens Cleaning Devices
The Dry Eye Foundation is warning consumers against the following products for contact lens cleaning and disinfection:
ReO2 Gen 6 Soft Contact Lens Cleaner
ReO2 Travel Contact Lens Cleaner
ReO2 RGP Contact Lens Cleaner
ReO2 Scleral Lens Cleaner
ReO2 lens devices from 3N Eyecare
Concern #1: Lens disinfection claims.
ReO2 devices are advertised as complete cleaning systems that can sterilize lenses using saline solution or tapwater.
However, 3N Eyecare’s regulatory documents for ReO2 state that the product is exclusively for protein removal and must be used in conjunction with one of two specified multi-purpose solutions that are approved for disinfection.
In other words, the 3N device performs an extra cleaning step, but cannot replace daily disinfection with a multi-purpose or disinfection solution.
Concern #2: Unsafe product instructions.
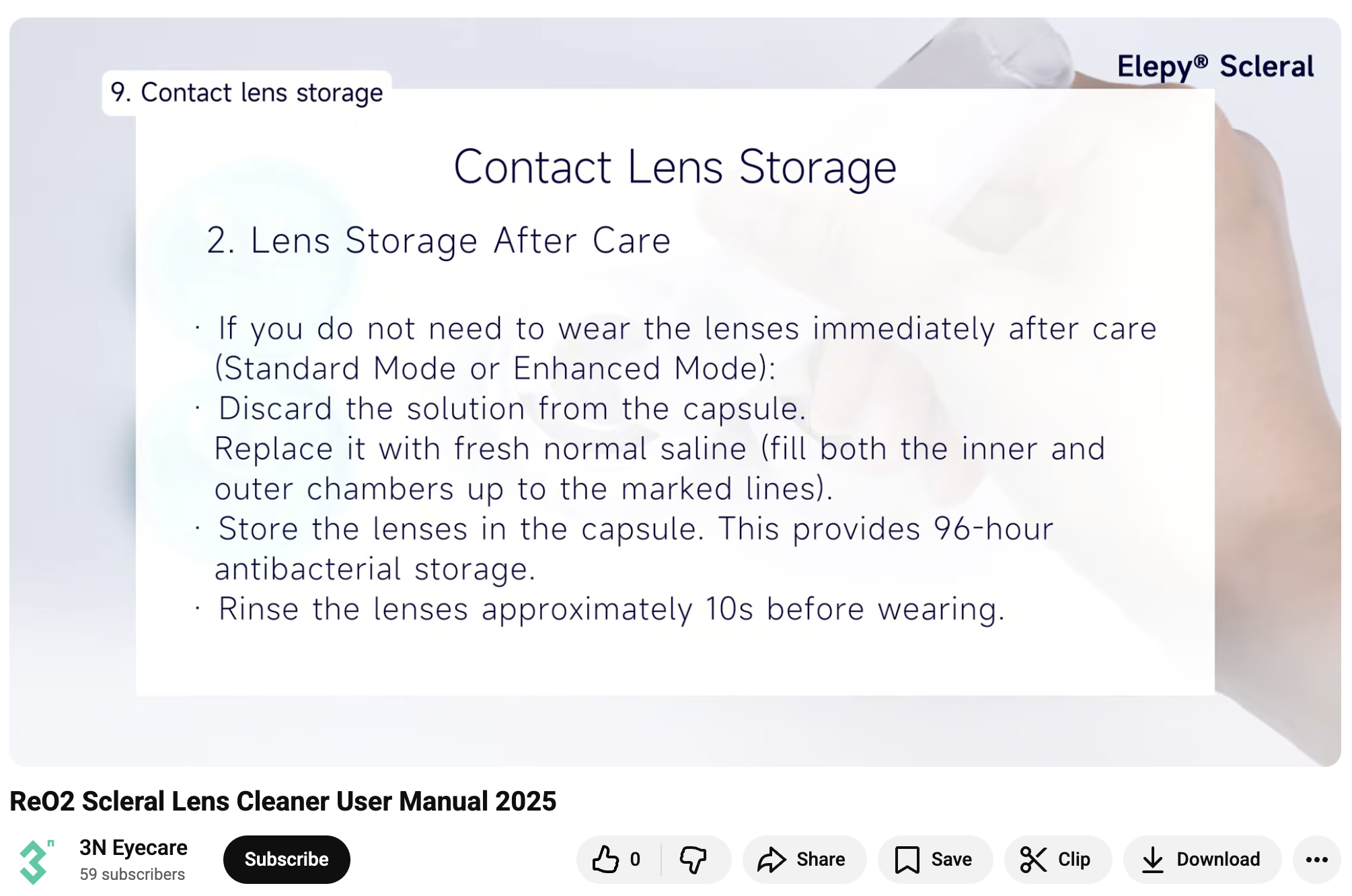
Storing contact lenses incorrectly increases the risk of serious eye infections, including Acanthamoeba Keratitis.
3N’s YouTube user manual for ReO2 indicates that storing lenses in their capsule filled with “normal saline”’ (i.e. preservative-free unbuffered saline solution) is appropriate for multi-day sterile storage and even antibacterial.
Concern #3: False claims about FDA approval of the ReO2 scleral lens device.
3N has a device clearance for deproteinization of soft lenses only.
3N Eyecare has falsely claimed or implied in their advertising to eye care providers that their scleral lens device is FDA approved or cleared for deproteinization and disinfection of scleral lenses.
Search results in FDA’s 510(k) database
Subject lines from 3N’s 510(k) document
From a trade show in January 2026